Glynase XL-10 – Strip of 10 Tablets
Amazon.in Price: Original price was: ₹20.63.₹17.74Current price is: ₹17.74. (as of 30/12/2025 23:50 PST- Details)
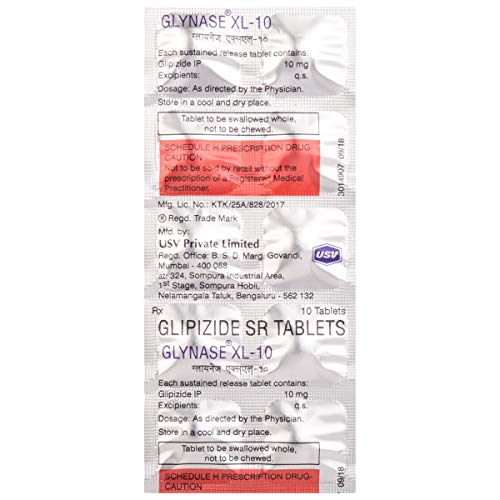
Composition: Each sustained release tablet contains: Glimepiride IP 10 mg, Excipients: q.s.
This product will have minimum 3 months expiry at the time of order dispatch.
Dosage: As directed by the Physician.
Description
Composition: Each sustained release tablet contains: Glimepiride IP 10 mg, Excipients: q.s. Dosage: As directed by the Physician.
Composition: Each sustained release tablet contains: Glimepiride IP 10 mg, Excipients: q.s.
This product will have minimum 3 months expiry at the time of order dispatch.
Dosage: As directed by the Physician.
Storage Instructions: Store in a cool and dry place.
Manufactured/Marketed By: Usv Private Limited
Schedule H prescription drug – Caution: Not to be sold by retail without the prescription of a Registered Medical Practitioner.
Tablet to be swallowed whole, not to be chewed.
Additional information
| Manufacturer | |
|---|---|
| Country of Origin | |
| Product Dimensions | |
| Packer | |
| Item Weight | |
| Item Dimensions LxWxH | |
| Net Quantity | |
| Included Components | |
| Generic Name | |
| Brand |
Only logged in customers who have purchased this product may leave a review.
General Inquiries
There are no inquiries yet.











Reviews
There are no reviews yet.